Ultravision Technology Up To 23x Better
Ultravision is the world's first system that eliminates surgical smoke without the need for gas exchange.
The revolutionary new system improves the surgical experience by providing surgical smoke control at the point of origin. In both minimally invasive and open surgery Ultravision delivers advanced visualization, unparalleled bioaerosol control and minimizes workflow disruptions caused by the need to manage surgical smoke. In addition, for MIS surgery, Ultravision benefits patients by delivering the ability to maintain a stable pneumoperitoneum in standard and low pressure surgery, whilst minimizing patient carbon dioxide exposure.
Ultravision For Open Surgery
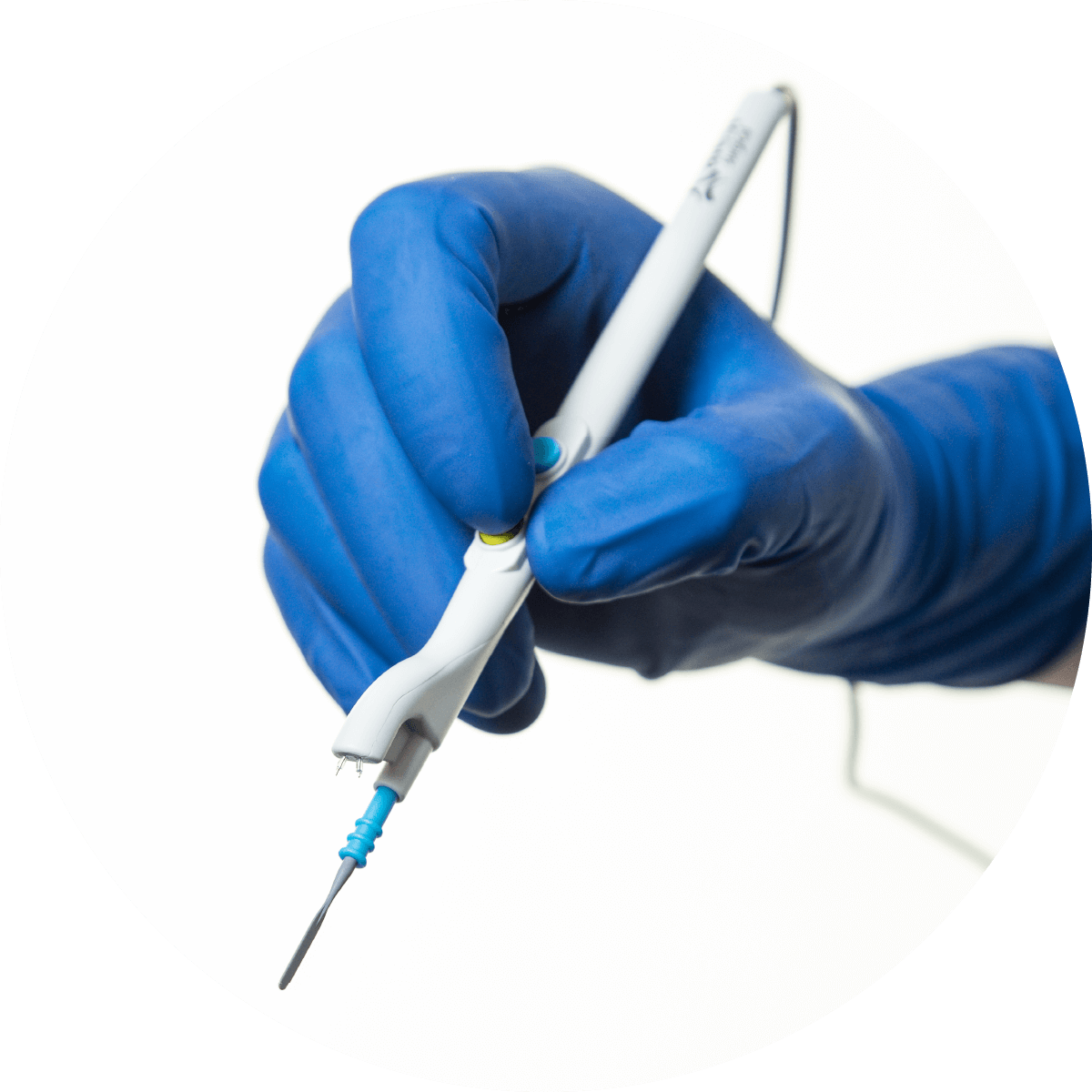
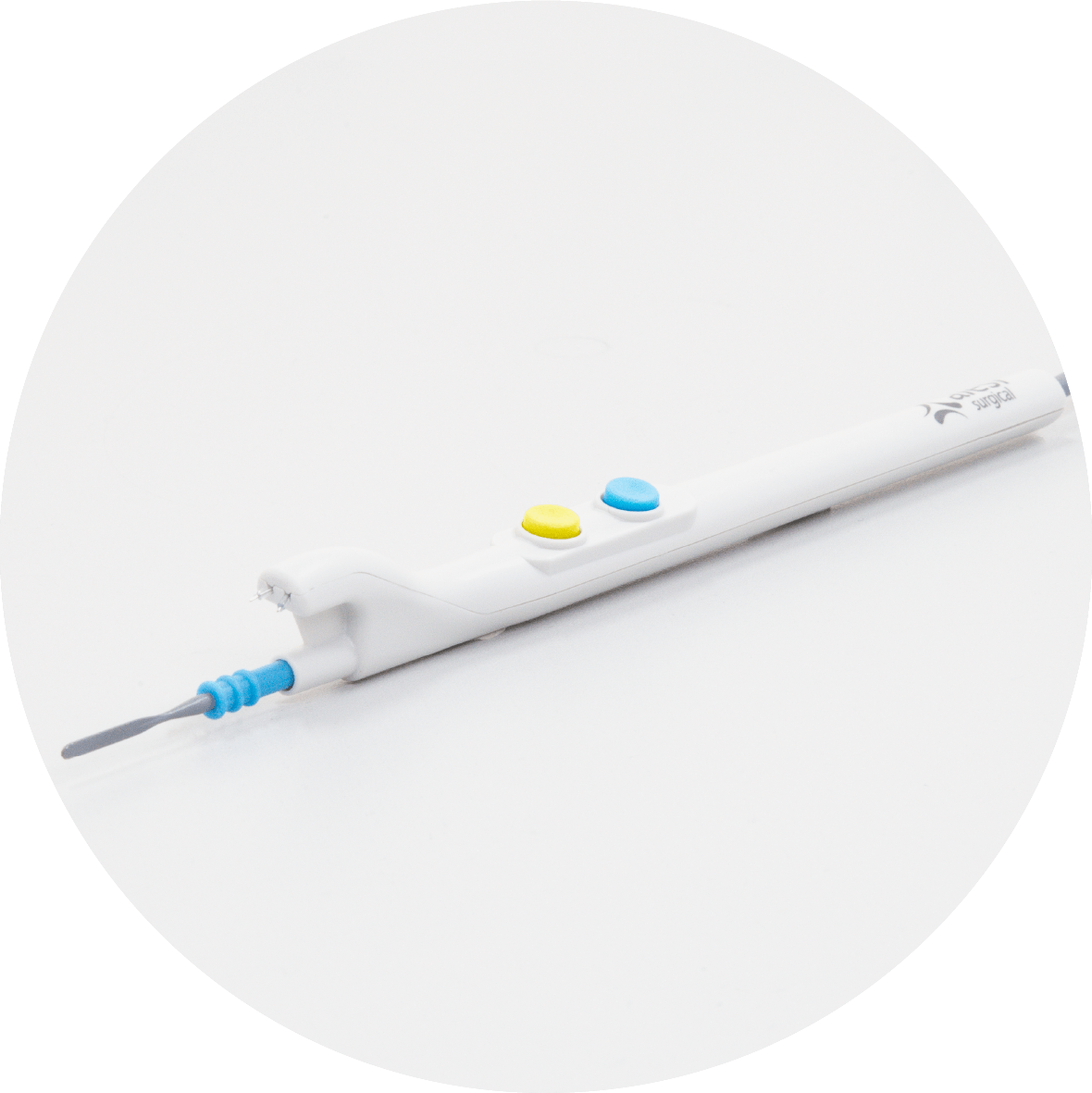
The only diathermy pencil that silently manages surgical smoke at the point of origin.
The IonPencil is a world first, offering silent, vacuum-free management of surgical smoke. Its slimline design offers excellent surgical smoke management without the need for cumbersome, bulky, noisy smoke evacuator systems.
Ultravision For
Laparoscopic Surgery
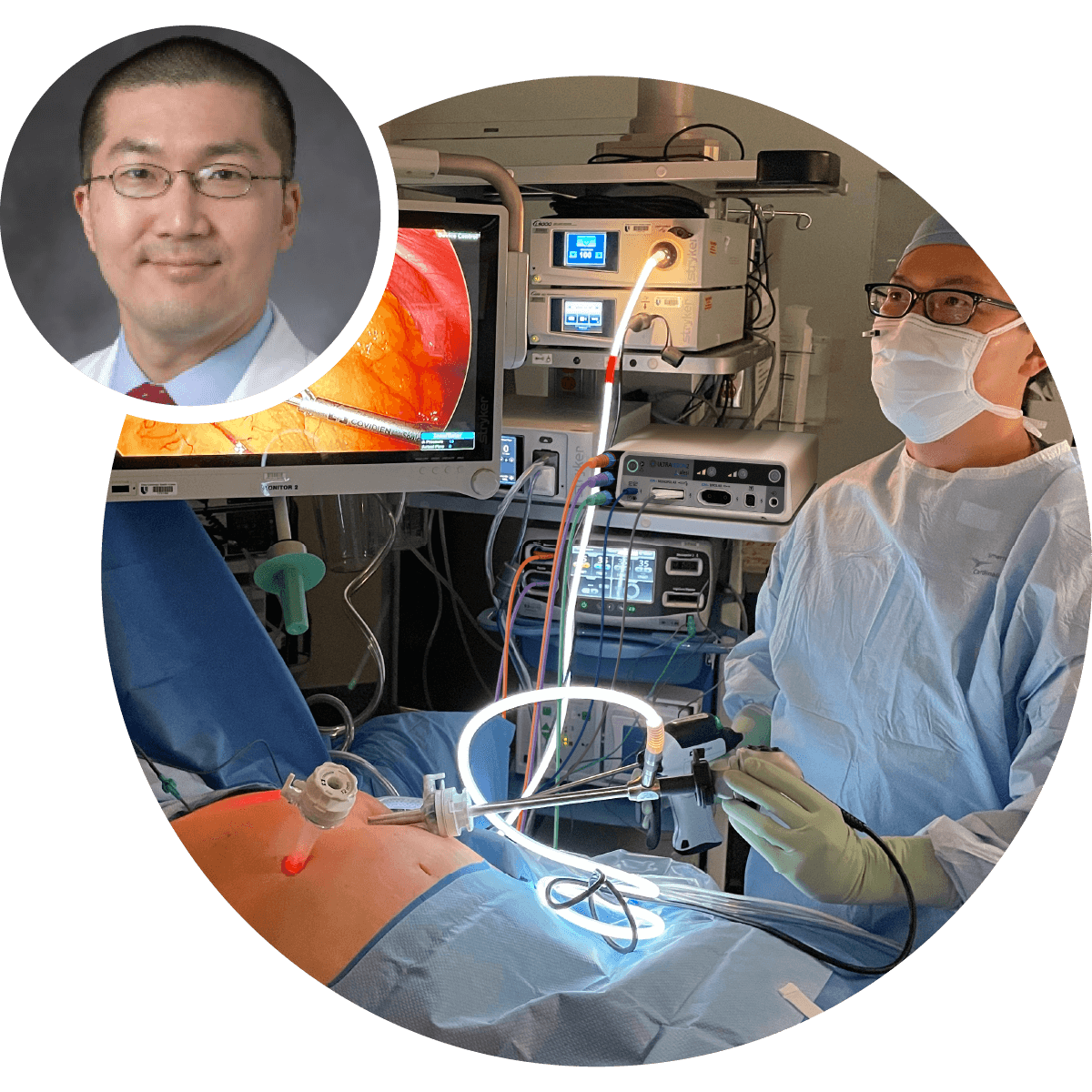
The only advanced visualization solution that prevents surgical smoke from entering the OR whilst minimizing patient carbon dioxide exposure during laparoscopic surgery.
Ultravision's unique mode of action delivers unparalleled bioaerosol control, advanced visualization, stable pneumoperitoneum in standard and low pressure surgery, and minimizes workflow disruptions caused by the need to manage surgical smoke.
The unique mode of action of Ultravision™
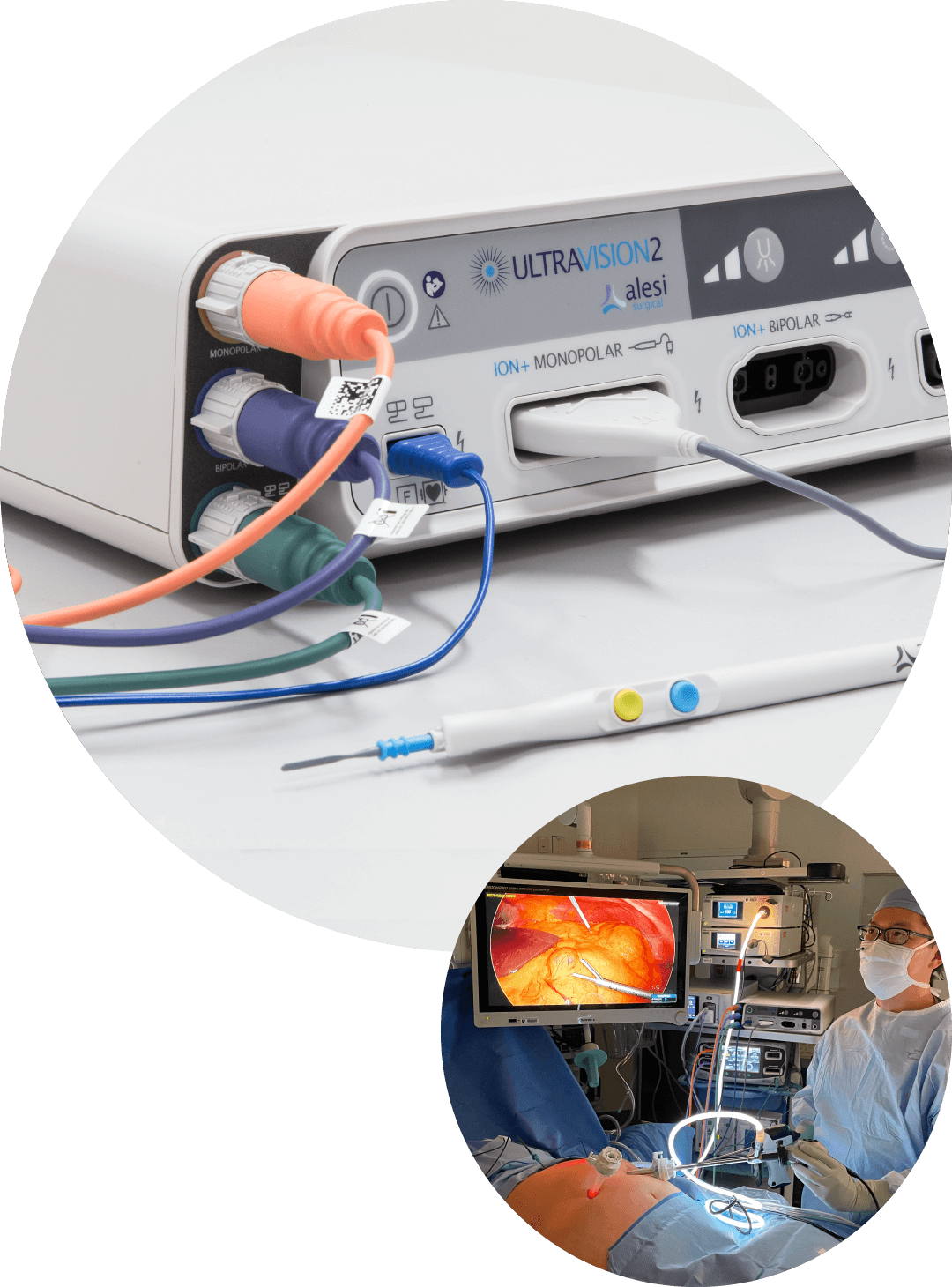
Ultravision™ technology is the only one in its field that uses the proven process of electrostatic precipitation to eliminate surgical smoke.
This unique mode of action delivers "advanced vizualisation": a continuously clear visual field, reduced camera cleaning, and stable pneumoperitoneum. Ultravision also provides best-in-class control of bioaerosols, being 23-times more effective than smoke evacuators in minimising the release of bioaerosols into the OR during surgery.*
*Source: Buggisch et al (2020)
How Electrostatic Precipitation works in Laparoscopic Surgery
The Ionwand™ creates negatively charged gas ions in the abdominal cavity, which move towards the “positive” patient tissue.
As the ions move, they collide with water vapour and particulate matter and draw them away from the surgical site. Particles land, and the electrical charge flows back to the generator.
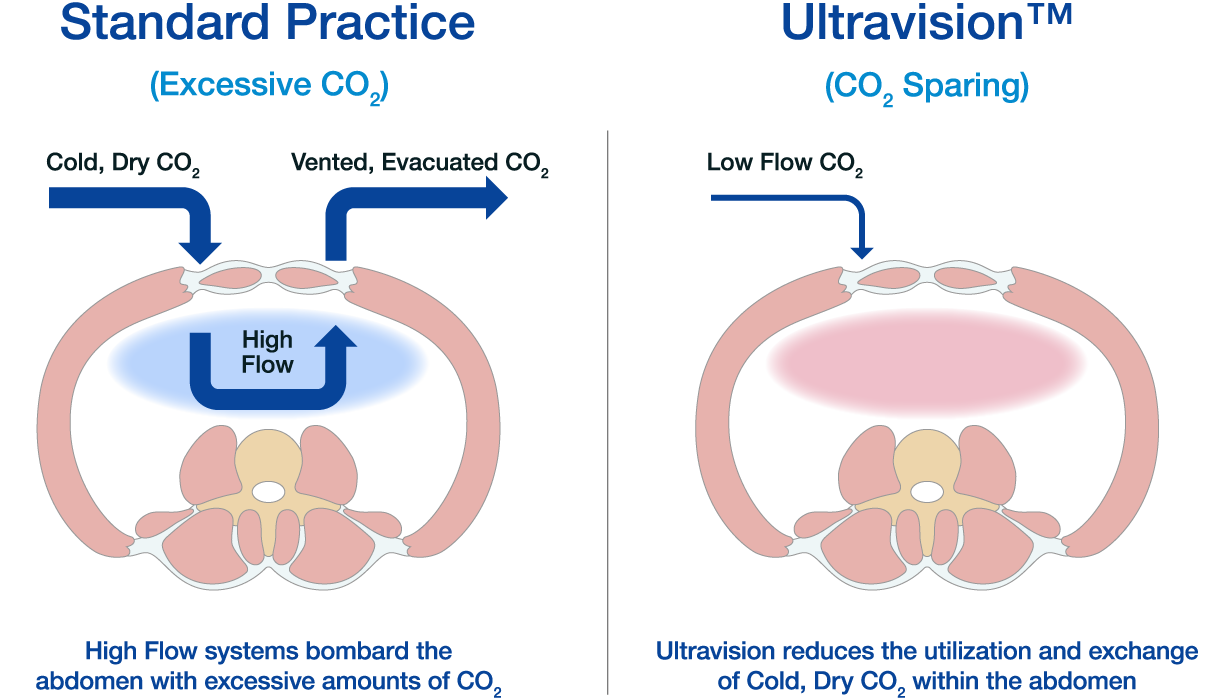
The Ultravision™ Differentiation & Paradigm Shift
Standard smoke evacuation systems require a constant flow of CO2 to clear the visual field from surgical smoke. This demands excessive use of CO2, with potentially negative effects on the patient. The electrostatic precipitation method enables several potential benefits including, improved vizualisation, low flow surgery, reduced CO2 usage, reduced exposure to surgical smoke in the OR and improved patient outcomes.
Questions and Answers
The rate of adoption of surgical smoke legislation is accelerating as the short- and long-term exposure risks become better understood. In 2018, only 1 state had enacted legislation. Just six years later and now over twenty states have either enacted, or are in the process of enacting legislation mandating the use of smoke management products in the OR.
As of July 2024, 18 states have enacted legislation requiring hospitals to provide surgical smoke management equipment, with a further four pending.
The AORN organization is the best way to track progress, which has accelerated dramatically since the Covid pandemic:
https://www.aorn.org/get-involved/government-affairs/policy-agenda/surgical-smoke-free-or/smoke-bills
In Europe, EORNA is proactively lobbying for similar legislation to be introduced in Europe.